AYVAKIT
The body factory
Concepting / Copywriting / Production
To help patients understand how AYVAKIT works, we created “The Body Factory,” a helpful metaphor that showcases what happens inside the body when someone has ISM and takes AYVAKIT to treat their symptoms.
AGENCY: FCB Chicago
OVERVIEW
In May 2024, AYVAKIT launched as the first and only FDA-approved treatment for adults with indolent systemic mastocytosis (ISM). In 2025, we created “The Body Factory” to help patients understand how this treatment works differently from over-the-counter medications by targeting the source of ISM to reduce symptoms.


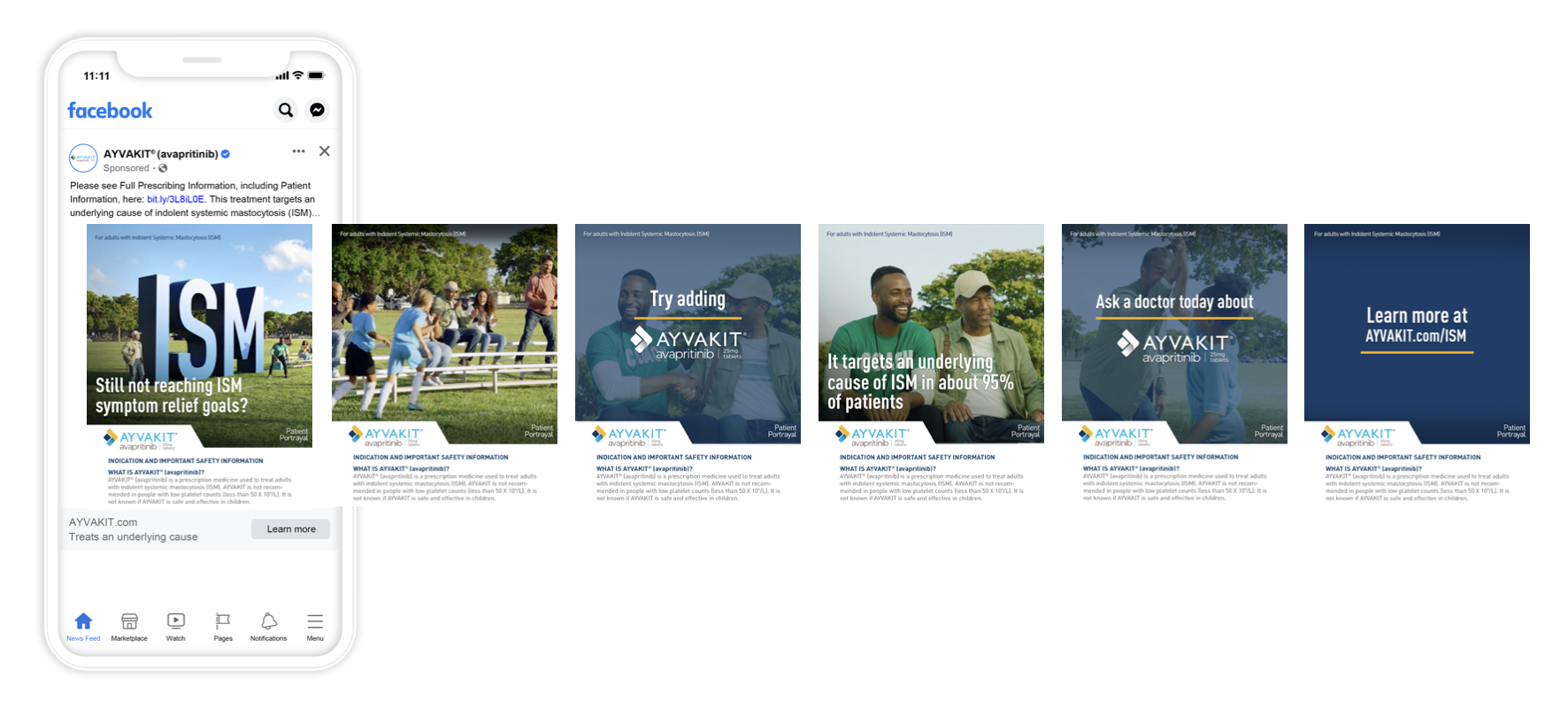
SOCIAL
We figured out how to reach our patient audience on social media creatively (and within regulatory guidelines) and help them understand how AYVAKIT can help treat this rare disease.